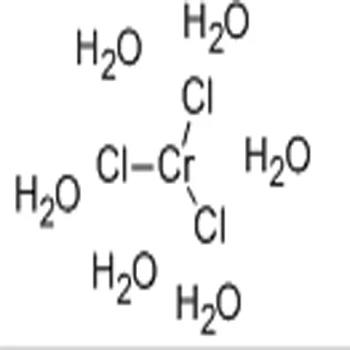  
Neither ILO nor WHO nor the European Commission shall be responsible for the interpretation and use of the information contained in this material. The molar mass of chromium(III) chloride hexahydrate is 266.45 g/mol. of a 'cocktail' composed of magnesium chloride hexahydrate, hydroxocobalamin. The molar mass of chromium(III) chloride is 158.36 g/mol. The published material is being distributed without warranty of any kind, either expressed or implied. magnesium, vitamin C, a multivitamin/min- eral formula, liquid garlic. Melting points vary depending on crystal structure.Īll rights reserved. The substance is toxic to aquatic organisms. Browse Chromium(III) chloride hexahydrate and related products at MilliporeSigma. to Safe Work Australia - Code of Practice Chromium(III) chloride hexahydrate 97, p.a. TLV: (as Cr(III), inhalable fraction): 0.003 mg/m 3, as TWA A4 (not classifiable as a human carcinogen) (DSEN) (RSEN). Synonyms: Chromium trichloride hexahydrate, Hexaaquachromium(III) chloride. Name of substance Chromium(III) chloride hexahydrate Molecular formula ClCr 6 HO Molar mass 266.4 g/mol CAS No 1 Safety data sheet Safety data sheet acc. Repeated or prolonged contact may cause skin sensitization. The substance is irritating to the respiratory tract.Ī harmful concentration of airborne particles can be reached quickly when dispersed.Įffects of long-term or repeated exposure Although it is ionic, the solid state structure is kinetically inert so that anhydrous CrCl 3 is surprisingly reluctant to dissolve in water. With the financial assistance of the European Commission. Chromium(III) chloride (also called chromic chloride) is a violet coloured solid with the formula CrCl 3. Prepared by an international group of experts on behalf of ILO and WHO, If appropriate, moisten first to prevent dusting. 266.45 GB5450000 Solubility Parameter SOLUBLE 9BZ4J5LYXjs HEXAAQUACHROMIUM (III), CHLORIDE CHROMIC CHLORIDE, HEXAHYDRATE CHROMIUM TRICHLORIDE, HEXAHYDRATE. Sweep spilled substance into sealable containers. It is also used as a catalyst and as a precursor to many inorganic. Do NOT let this chemical enter the environment. The most common form of Chromium (III) Chloride is the dark green hexahydrate, CrCl3 6 H2O. Personal protection: particulate filter respirator adapted to the airborne concentration of the substance. 19:224 FORMULA INDEX The Formula Index, as well as the Subject Subject. Give one or two glasses of water to drink. a16': hexaammonium, hexahydrate, 19:140, 143 hexasodium, octadecahydrate. Rinse skin with plenty of water or shower.įirst rinse with plenty of water for several minutes (remove contact lenses if easily possible), then refer for medical attention.Ībdominal pain. Use local exhaust or breathing protection. In case of fire in the surroundings, use appropriate extinguishing media.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
